Фазовая диаграмма системы Mg-Nd
К оглавлению: Другие диаграммы (Others phase diargams)
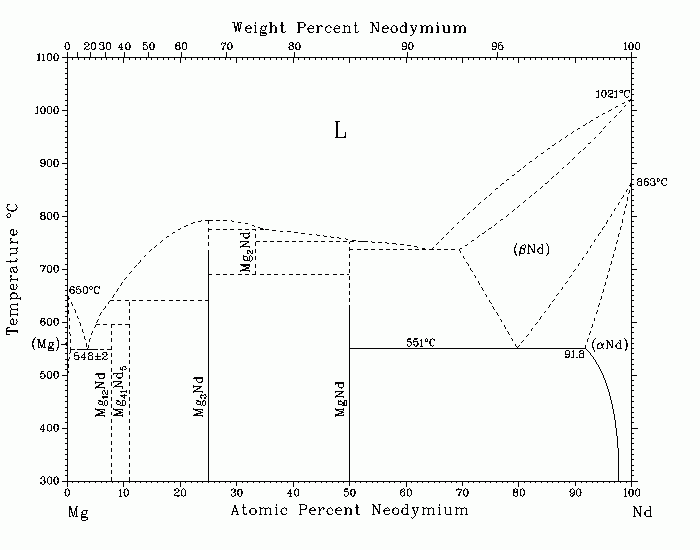
Mg-Nd (Magnesium-Neodymium) A.A. Nayeb-Hashemi and J.B. Clark The Mg-rich region of the Mg-Nd equilibrium phase diagram has been investigated extensively [58Sav, 58Tik, 62Rok, 63Dri]. However, the purity of the starting materials was often quite low, and alumina or corundum crucibles usually were used. Consequently, contamination of the melt by Mg and Nd reduction of the crucible materials could have occurred. For these reasons, the findings of these investigations are open to doubt. The assessed Mg-Nd phase diagram shows only the type and general configuration of the system. Further investigations are needed to establish the phase boundaries shown as dashed lines. [57Par] showed that the Mg-rich region is of the eutectic type. In the assessed diagram, the Mg-rich eutectic temperature can be placed with confidence at 548 с 2 C. The (Mg) liquidus, solidus, and solvus are shown as dashed lines, because the reports vary widely among investigators. The discrepancies of the solid solubilities reported are so large that a new, independent investigation is needed. [57Par] placed the maximum solid solubility of Nd in (Mg) at 0.1 at.% Nd at the eutectic temperature. Because of the higher-purity materials and the more reliable method used by [57Par], it is believed that the true solid solubility of Nd in (Mg) is closer to that reported by [57Par] than to the data of other investigators. [65Jos] determined that the maximum solid solubility of Mg in (aNd) at the eutectoid decomposition temperature of (bNd) (551 C) was 91.8 at.% Nd (8.2 at. % Mg). There is controversy concerning the stoichiometry of the compound in equilibrium with (Mg). Because the presence of the Mg-rich compounds Mg12RE and Mg41RE5 in the Mg-La, Mg-Ce, and Mg-Pr systems has been established, and because of the reliability of the X-ray results of [63Evd], [64Kri], [64Las], and [69Kri], it is possible that there are at least two Mg-rich compounds in the Mg-Nd system-possibly Mg12Nd, which is in equilibrium with (Mg), and Mg41Nd5. [73Bus] and [76Bus] investigated the magnetic properties of MgRE and Mg3RE compounds, respectively, in the temperature range 4 to 300 K. According to [ 73Bus], the temperature dependence of the magnetization of MgNd gives rise to a maximum, indicative of a paramagnetic-antiferromagnetic transition. The maximum N‚el temperature of MgNd is 48 K, and the asymptotic Curie temperature (qp) is -14 K. Although [76Bus] found that Mg3Nd does not show any magnetic ordering and that qp for Mg3Nd from linear extrapolation is -124 K, [82Gal] reported qp as -58 K (see also [81Gal]). [78Bus] studied the magnetic properties of Mg2RE compounds in the temperature range 1.5 to 300 K and the magnetic field up to 18 kOe. According to [78Bus], most of the Mg2RE compounds order ferromagnetically at temperatures "well below 100 K," except for Mg2La, Mg2Yb, and Mg2Y, which show Pauli paramagnetism; Mg2Sm and Mg2Eu order antiferromagnetically. The Curie temperature of Mg2Nd was found to be 26 K [78Bus], which is in fair agreement with 28.5 K, reported by [81Loi]. 57Par: J.J. Park and L.L. Wyman, WACD Tech. Rep. 57-504, 33 (1957). 58Sav: Ye.M. Savitskii, V.F. Terekhova, and I.A. Novikova, Zh. Neorg. Khim., 3( 9), 2138-2142 (1958) in Russian. 58Tik: N.M. Tikhova and L.A. Afanas'eva, Metalloved. Obrab. Met., (3), 38-41 ( 1958) in Russian; TR: Brutcher Transl. No. 4166. 59Ian: A. Iandelli, Natl. Phys. Lab., Great Britain, Proc. Symp. No. 9, Vol. I, Paper 3F, 11 p (1959). 60Ian: A. Iandelli, Atti. Accad. Naz. Lincei Rend., 29, 62-69 (1960) in Italian. 62Rok: L.L. Rokhlin, Izv. Akad. Nauk SSSR, Otd. Tekh. Nauk, Met. Toplivo, (2), 126-130 (1962) in Russian; TR: Russ. Met. Fuels, (2), 98-100 (1962). 63Dri: M.E. Drits, Z.A. Sviderskaya, and L.L. Rokhlin, Met. Metalloved., Fiz. Khim. Metody. Issled., (12) 143-151 (1963) in Russian. 63Evd: V.I. Evdokimenko and P.I. Kripyakevich, Kristallografiya, 8, 186-193 ( 1963) in Russian; TR: Sov. Phys. Crystallogr., 8, 135-141 (1963). 64Kri: P.I. Kripyakevich and V.I. Evdokimenko, Problems of the Theory and Application of Rare Earth Metals, E.M. Savitskiy and V.F. Terekhova, Ed., Nauka, Moscow, 191-194 (1964). 64Las: N.F. Lashko and G.I. Morozova, Kristallografiya, 9, 269-270 (1964) in Russian; TR: Sov. Phys. Crystallogr., 9, 209-211 (1964). 65Ian: A. Iandelli and A. Palenzona, J. Less-Common Met., 9, 1-6 (1965). 65Jos: R.R. Joseph and K.A. Gschneidner, Jr., Trans. AIME, 223, 2063-2069 ( 1965). 66Cha: C.C. Chao and P. Duwez, J. Appl. Phys., 37(7), 2631-2632 (1966). 69Kri: P.I. Kripyakevich and V.I. Evdokimenko, Visn. L'vivs. Derzh. Univ., Ser. Khim., 3-6 (1969) in Russian. 72Pah: J.E. Pahlman and J.F. Smith, Metall. Trans., 3, 2423-2432 (1972). 73Bus: K.H.J. Buschow, J. Less-Common Met., 33, 239-244 (1973). 76Bus: K.H.J. Buschow, J. Less-Common Met., 44, 301-306 (1976). 78Bea: B.J. Beaudry and K.A. Gschneidner, Jr., Handbook on the Physics and Chemistry of Rare Earths, Vol. 1 - Metals, K.A. Gschneidner, Jr., and L. Eyring, Ed., North-Holland Publishing Co., Amsterdam, 173-232 (1978). 78Bus: K.H.J. Buschow, R.C. Sherwood, and F.S.L. Hsu, J. Appl. Phys., 49(3), 1510-1512 (1978). 81Gal: R.M. Galera, A.P. Murani, and J. Pierre, J. Magn. Magn. Mater., 23, 317- 322 (1981). 81Loi: A. Loidl, K. Knorr, M. Mulner, and K.H.J. Buschow, J. Appl. Phys., 52(3) , 1433-1438 (1981). 82Gal: R.M. Galera, J. Pierre, and J. Pannetier, J. Phys. F, Met. Phys., 12, 993-1003 (1982). Published in Phase Diagrams of Binary Magnesium Alloys, 1988, and Bull. Alloy Phase Diagrams, 9(5), Oct 1988. Complete evaluation contains 1 figure, 6 tables, and 37 references. Special Points of the Mg-Nd System
