Фазовая диаграмма системы Cu-Zn
К оглавлению: Другие диаграммы (Others phase diargams)
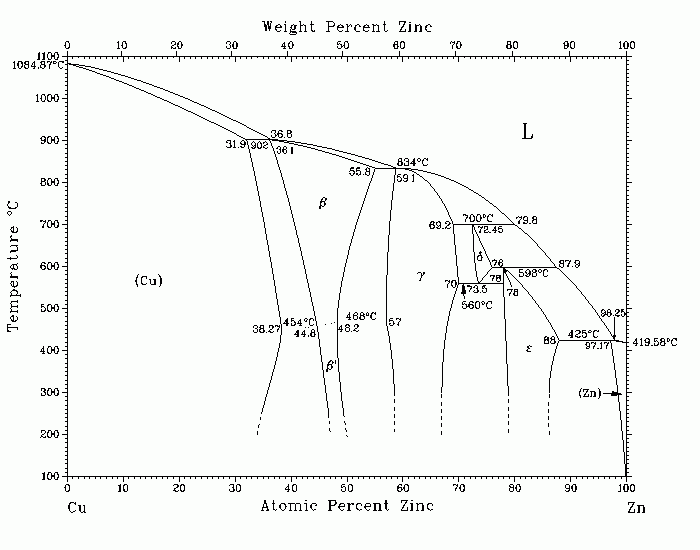
Cu-Zn (Copper-Zinc) A.P. Miodownik The assessed Cu-Zn phase diagram is modified from [Hansen], with review of the data of [21Hau], [22Ima], [25Gen], [27Bau], [29Rue], [35Sch], and [62Mas]. The assessed phase boundaries differ only marginally from those proposed by [36Han] and [44Ray]. Because there is little dispute about the phase boundaries, more recent research on this system has largely concentrated on elucidating finer detail concerning the structure of the individual phases, such as the possibility of short-range order in the a phase, the order-disorder transformation in b, polymorphism in g, and the defect structure of the d phase. As a result of interest in shape memory alloys, considerably more information is now also available on various martensitic transformations in the b phase. For the liquidus curve in the assessed diagram, preference has been given to the data of [27Bau], [29Rue], and [35Sch], which are accurate within с3 C [ 44Ray]. The liquidus curve between 54 and 100 at.% Zn was determined by [27Bau] . Numerous shear transformation products have been observed under various conditions; until recently, it was both difficult to assess the relationship between different observations and to maintain a sensible nomenclature [ Pearson2]. These difficulties have now been largely resolved, and the majority of observed metastable crystal structures can be placed within the classification scheme proposed by [86Ahl]. Among the earlier structures reported are an orthorhombic cell for an alloy with 38.3 at.% Zn quenched from 840 C [63Jol], various metastable tetragonal phases [59Hor], and monoclinic and triclinic products when cooling the b› phases to liquid nitrogen temperatures [62Kun]. Following the classification of [86Ahl], the most common martensitic structure is best described by a stacking sequence described as 9R or 18R. In alloys containing 37.6 to 37.7 at.% Zn (corresponding to lower electron concentrations), [74Cor] reported a structure consisting of 3R twins, and [ 82Kaj] have found 3R and 9R plates, which are fcc-like structures consistent with the proximity of the stable a phase. By contrast, at higher electron concentrations, [61Rao] found a hexagonal product in alloys containing 39.5 to 40.5 wt.% Zn, which is consistent with the variation in stacking fault energy with concentration, and the relative metastabilities of various close-packed phases [74War, 85Mio, 86Ahl]. This also confirms the earlier statement of [62Mas] concerning the limitations of too strict an interpretation of the Hume-Rothery rules in such systems. Additional structural changes can be produced by deformation [57Hor], and [ 74Arn], and still further variants are observed as surface martensites [83Lov]. According to the assessed diagram, the most Cu-rich alloy that can be heat- treated in the b phase field contains 36.2 at.% Zn, but [85Euc] has pointed out that, in principle, alloys with higher Ms temperatures could be obtained from alloys containing metastable b via rapid solidification. It should be noted that the absolute values of the Ms temperature are affected by the degree or order in the parent phase, and hence by quenching velocity and prior aging treatments [79Rap, 80Sch]. Many of these structures exhibit thermoelastic behavior and low energy interfaces, so it is possible to obtain reasonable estimates of the relative energies and entropies of various competing martensites, their parent phases, and related equilibrium phases [79Cha, 80Cha, 86Ahl]. Such treatments confirm the small free energy differences that exist between various structures, which is consistent with observed difficulties in obtaining reproducibility. The fact that martensitic structures inherit order from the parent phase means that varying degrees of order in the parent phase can directly account for observed distortions such as a shift from orthorhombic to monoclinic symmetry [ 77Cah, 86Ahl]. The experimental observation of a set of ordered close-packed martensites also implies that such ordered structures are relatively stable in their own right. This is confirmed by the fact that the observed variation of Ms temperature with Zn concentration cannot be matched by calculations without assuming ordering in the product phase [79Cha, 80Cha], which in turn confirms the likelihood for the presence of ordering in the a phase at low temperatures. 21Hau: J.L. Haughton and K.E. Bingham, Proc. R. Soc. (London) A, 99, 46-69 ( 1921). 22Ima: H. Imai, Sci. Rep. T“hoku Imp. Univ., 11(1), 313-332 (1922). 25Gen: R. Genders and G.L. Bailey, J. Inst. Met., 33, 213-223 (1925). 27Bau: O. Bauer and M. Hansen, Z. Metallkd., 19, 423-434 (1927). 29Rue: R. Ruer and K. Kremers, Z. Anorg. Chem., 184, 193-231 (1929). 35Sch: J. Schramm, Metallwirtschaft., 14, 995-1001, 1047-1050 (1935). 36Han: Handbook of Binary Alloys, M. Hansen, Springer-Verlag, Berlin, 652-672 ( 1936). 44Ray: G.V. Raynor, Annotated Equilibrium Diagram Series, No. 3., Institute of Metals, London (1944). 54Tit: A.L. Titchener and M.B. Bever, Trans. AIME, 200, 303-304 (1954). 57Hor: E. Hornbogen, A. Segmuller, and G. Wassermann, Z. Metallkd., 48, 379- 384 (1957). 59Hor: E. Hornbogen, Z. Metallkd., 50, 70-74 (1959). 61Rao: S.S. Rao and T.R. Anantharaman, Naturwissenschaften, 48, 712-716 (1961). 62Kun: G. Kunze, Z. Metallkd., 53, 329-341 (1962). 62Mas: T.B. Massalski and H.W. King, Acta Metall., 10, 1171-1181 (1962). 63Jol: W. Jolley and D. Hull, J. Inst. Met., 92, 129-135 (1963/64). 64Pop: H. Pops and T.B. Massalski, Trans. AIME, 230, 1662-1668 (1964). 74Arn: W. Aredo and M. Ahlers, Acta Metall., 22, 1475-1480 (1974). 74Cor: L. Cornelis and C.M. Wayman, Acta Metall., 22, 291-301 (1974). 74War: H. Warlimont and L. Delacy, Progress in Materials Science, Vol. 18, C. Christian and T.B. Massalski, ed., New York, 1-157 (1974). 77Cah: J.W. Cahn, Scr. Metall., 11, 82-82 (1977). 79Cha: L. Chandrasekaran and A.P. Miodownik, Proceedings of the International Conference on Martensitic Transformations (ICOMAT), Cambridge, MA, W. Owen, G. B. Olson, and P. Clapp, ed., 584-589 (1979). 79Rap: R. Rapacioli and M. Ahlers, Acta Metall., 27, 777-784 (1979). 80Cha: L. Chandrasekaran, Ph.D. Thesis, University of Surrey, chap. 5, 71-84 ( 1980). 80Sch: D. Schofield and A.P. Miodownik, Met. Technol.,6(4), 167-173 (1980). 82Kaj: S. Kajiwara and T. Kiknclin, Acta Metall., 30, 589-598 (1982). 83Lov: F.C. Lovey and M. Chandrasekaran, Acta Metall., 31, 1919-1927 (1983). 85Euc: S. Eucken and E. Hornbogen, Rapidly Quenched Metals, S. Steep and H. Warlimont, ed., Elsevier, New York 1429-1434 (1985). 85Mio: A.P. Miodownik, J. Less-Common Met., 114(1), 81-88 (1985). 86Ahl: M. Ahlers, Prog. Mater. Sci., 30, 135-186 (1986). Submitted to the APD Program. Complete evaluation contains 6 figures, 5 tables, and 115 references. Special Points of the Cu-Zn System
