Фазовая диаграмма системы Al-Fe
К оглавлению: Другие диаграммы (Others phase diargams)
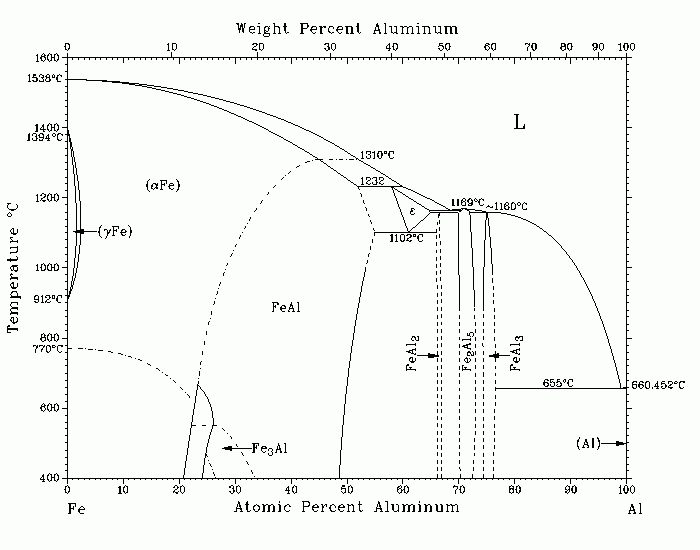
Al-Fe (Aluminum-Iron) U.R. Kattner The assessed phase diagram for the Fe-Al system has not yet been determined completely with precision. Although the existence of the equilibrium intermediate phases appears to be reliably established, refinement of the reaction temperatures and phase boundaries is needed. The assessed diagram is based on [25Dix], [27Gwy], [60Lee], [67Roc], [75All], [80Kos], [80Sch], [86Gri] , and [87Wac]. The equilibrium phases are the liquid, L; the (aFe) solid solution based on bcc Fe, where up to 45 at.% Al dissolves in (aFe) when ordering starts to occur; the (gFe) solid solution based on fcc Fe, where up to 1.3 at.% Al dissolves in (gFe); FeAl, which forms through ordering of (aFe); Fe3Al, which forms through a second-order phase transformation from FeAl; the e phase, with a solution range extending from ~58 to ~65 at.% Al; FeAl2, with a narrow range of solubility from 66 to 66.9 at.% Al; Fe2Al5, with a range of solubility from ~70 to ~73 at.% Al; FeAl3, with a range of solubility from 74.5 to 76.6 at.% Al; and the (Al) solid solution, with a solubility of up to 0.03 at.% Fe in ( Al). From rapid cooling of Al-rich liquid alloys the occurrence of two metastable compounds is known: FeAl6, which can be obtained with moderate quenching rates, and Fe2Al9, which only can be obtained with high quenching rates. The metastable eutectic involving liquid, (Al), and FeAl6 was determined by [82Ric] . From extrapolation of ternary equilibria involving these phases, [83Per] showed that the temperature of the metastable eutectic involving liquid, (Al), and Fe2Al9 lies below the temperature of the L = (Al) + FeAl6 metastable eutectic. [69Ton] reported that up to 4.4 at.% Fe can be obtained through rapid quenching of supersaturated (Al) solutions. 25Dix: E.H. Dix, Proc. ASTM, 25(2), 120-136 (1925). 27Gwy: A.G.C. Gwyer and H.W.L. Phillips, J. Inst. Met., 38, 29-83 (1927). 60Lee: J.R. Lee, J. Iron Steel Inst., 194, 222-224 (1960). 67Roc: P. Rocquet, G. Jegaden, and J.C. Petit, J. Iron Steel Inst., 205, 437- 441 (1967). 75All: S.M. Allen and J.W. Cahn, Acta Metall., 23, 1017-1026 (1975). 80Kos: W. Koester and T. Goedecke, Z. Metallkd., 71, 765-769 (1980) in German. 80Sch: E. Schuermann and H.-P. Kaiser, Arch. EisenhЃttenwes., 51, 325-327 ( 1980) in German. 82Ric: J. J. Richmond, J.H. Perepezko, S.E. LeBeau, and K.P. Cooper, Rapid Solidification Processing: Principles and Technologies III, (1982); Washington, DC, NBS, quoted by [83Per]. 83Per: J.H. Perepezko and W.J. Boettinger, Mater. Res. Soc. Symp. Proc., 19, 223-239 (1983). 86Gri: A. Griger, V. Stefaniay, and T. Turmezey, Z. Metallkd., 77, 30-35 (1986) . 87Wac: E. Wachtel and J. Bahle, Z. Metallkd., 78, 229-232 (1987) in German. Submitted to the APD Program. Complete evaluation contains 2 figures, 2 tables, and 19 references. Special Points of the Fe-Al System
